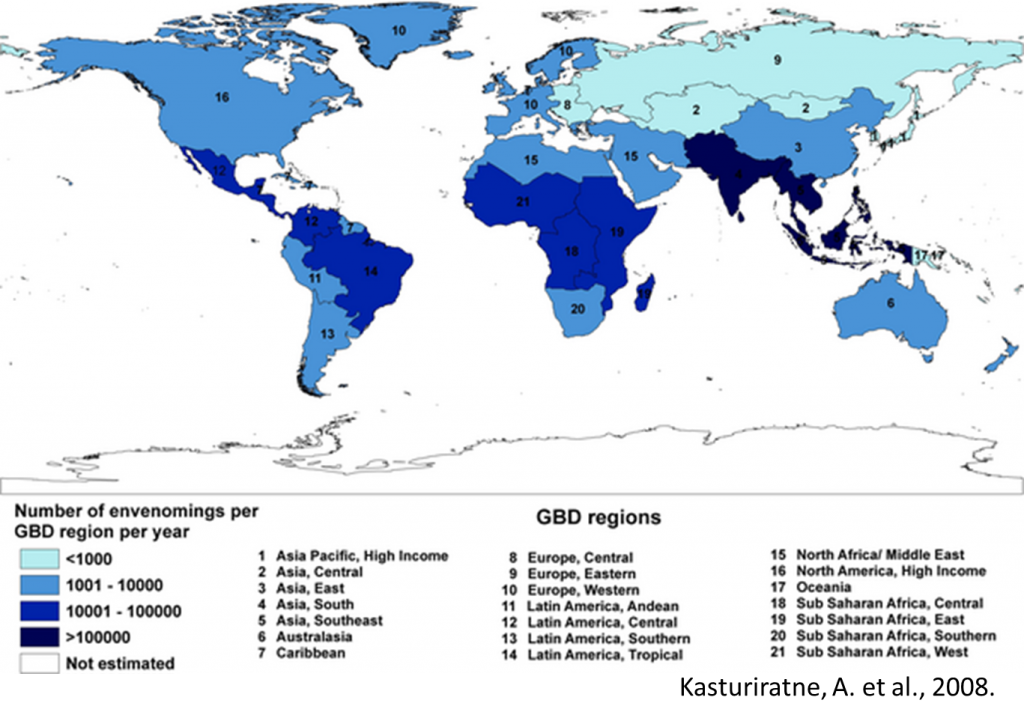
 Snakebites kill more than 100,000 people annually, making snakes the second deadliest animals (after mosquitos), with many fold that many people suffering permanent disabilities from snake venom. Most of the victims are in Africa, Asia and Latin America, often in countries with poor access to medical resources. Snake venoms are complex and contain various toxins including proteases and nucleases that destroy affected tissues. Some venoms target nervous tissues, some are anticoagulants, and some cause tissue necrosis.
Snakebites kill more than 100,000 people annually, making snakes the second deadliest animals (after mosquitos), with many fold that many people suffering permanent disabilities from snake venom. Most of the victims are in Africa, Asia and Latin America, often in countries with poor access to medical resources. Snake venoms are complex and contain various toxins including proteases and nucleases that destroy affected tissues. Some venoms target nervous tissues, some are anticoagulants, and some cause tissue necrosis.
Antivenoms (aka antivenins) are the only effective treatment for snakebite. Antivenoms are produced essentially the same way now as they were when they were developed in the late 1800s by Albert Calmette. Snakes are milked to obtain their venom which is diluted and injected into a horse or other animal. The animal produces polyclonal antibodies against the various components in the venom. When injected into a snakebite victim, the donor animal’s antibodies bind to and inactivate the venom toxins.
Unfortunately, antivenoms are expensive. Their production demands a supply of snake venom, which can only be obtained by cultivating venomous snakes, and a facility for housing the antibody-producing animals. Furthermore, the prepared antivenoms require refrigeration. In spite of there being an ongoing demand for antivenoms, many producers are finding them unprofitable and are curtailing their production. The World Health Organization has described the short supply of antivenom is a “neglected public health issue”. Alternative approaches to antivenom production are being explored, including the development of synthetic venoms to avoid the need to maintain colonies of venomous snakes.
Traditional medicines include descriptions of many plants with antivenom properties, but few of their mechanisms have been identified. One of these, velvet bean (Mucuna pruriens), seems to act both directly and indirectly against the venom of the saw-scaled viper,  Echis carinatus. Previously, a protein was identified from Mucuna pruriens seed extracts that appears to have protease inhibitor activity. As many snake venom toxins are proteases, this activity could explain the protective activity of the seeds. However, traditional wisdom says that, “when two seeds are swallowed, they protect the individual for a year against snakebites,” a claim that is hard to align with their functioning simply as protease inhibitors.
Echis carinatus. Previously, a protein was identified from Mucuna pruriens seed extracts that appears to have protease inhibitor activity. As many snake venom toxins are proteases, this activity could explain the protective activity of the seeds. However, traditional wisdom says that, “when two seeds are swallowed, they protect the individual for a year against snakebites,” a claim that is hard to align with their functioning simply as protease inhibitors.
Other studies have raised that possibility that the Mucuna pruriens mode of action is through an immunological function. Specifically, there appears to be cross-reactivity between proteins in the seed extract and proteins in the Echis carinatus venom. A new study by Kumar et al in the Journal of Biological Chemistry identifies a specific protein, MP-4, as that which reacts with antibodies raised against Echis carinatus venom, thus strengthening the idea that the protective function is immunological. Furthermore, mice immunized with MP-4 show significantly higher rates of survival than unimmunized mice when challenged with Echis carinatus venom. Thus, there may be merit to the idea that seed consumption can provide long-term protection against snakebite, through the stimulation of antibodies that cross-react with venom toxins.
Given that Mucuna pruriens seed extracts have several bioactive compounds including L-DOPA and have been recommended for treatment of various ailments from Parkinson’s to infertility and depression, wide-scale prophylactic consumption of Mucuna pruriens seeds may not be desirable. Identifying one or a few components that alone confer protection opens the door to an inexpensive, accessible strategy to combat the scourge of snakebite.
References
Arnold, C. (2016). Synthetic biology tackles global antivenom shortage. Nature 532: 292.
Guerranti, R., Aguiyi, J.C., Neri, S., Leoncini, R., Pagani, R. and Marinello, E. (2002). Proteins from Mucuna pruriens and enzymes from Echis carinatus venom: Characterization and cross-reactions. J. Biol. Chem.. 277: 17072-17078.
Hope-Onyekwere, N.S., Ogueli, G.I., Cortelazzo, A., Cerutti, H., Cito, A., Aguiyi, J.C. and Guerranti, R. (2012). Effects of Mucuna pruriens protease inhibitors on Echis carinatus venom. Phytotherapy Res.. 26: 1913-1919.
Houghton, P.J. and Osibogun, I.M. (1993). Flowering plants used against snakebite. J. Ethnopharmacol. 39: 1-29.
Hsu, J. (2015). Defanging snakebites. Scientific American 313: (December) 14 – 16.
Kasturiratne, A., Wickremasinghe, A.R., de Silva, N., Gunawardena, N.K., Pathmeswaran, A., Premaratna, R., Savioli, L., Lalloo, D.G. and de Silva, H.J. (2008). The global burden of snakebite: A literature analysis and modelling based on regional estimates of envenoming and deaths. PLoS Med. 5: e218.
Kumar, A., Gupta, C., Nair, D.T. and Salunke, D.M. (2016). MP-4 contributes to snake venom neutralization by Mucuna pruriens seeds through an indirect antibody-mediated mechanism. J. Biol. Chem. 291: 11373-11384.
Lampariello, L.R., Cortelazzo, A., Guerranti, R., Sticozzi, C., and Valacchi, G. (2012). The magic velvet bean of Mucuna pruriens. J. Tradit. Complement. Med. 2: 331–339.
Tan, N.H., Fung, S.Y., Sim, S.M., Marinello, E., Guerranti, R. and Aguiyi, J.C. (2009). The protective effect of Mucuna pruriens seeds against snake venom poisoning. J. Ethnopharmacol. 123: 356-358.
Warrell, D.A. (2010). Snake bite. The Lancet. 375: 77 – 88.
World Health Organization (2015). Snake antivenoms. Fact sheet No. 337.
Image credits: Mucuna pruriens: Public domain (Mucuna pruriens, a hand-coloured engraving after a drawing by Miss S. A. Drake (fl. 1820s-1840s), from the 24th volume of the Botanical Register (1838), edited by John Lindley), Echis carinatus: Shantanu Kuveskar.

If it really can prevent snake venom toxin then it’s a great news. Every day many many people died cause of snake bite over the world.
Thanks for sharing the information!