Chemical genetics is a powerful complement to conventional genetics. Rather than knocking out gene functions, protein (or other) functions can be perturbed through the addition of small molecules. One advantage is that several related proteins can be affected at the same time, avoiding the difficulties that come from genetic redundancy. Another is that treatment regimes can be transient, so even pathways required for viability can be examined (see more in Serrano et al, 2015, and Hicks and Raikhel, 2014).
A recent paper by et al describes a chemical genetics approach to study the process of lignification. The paper, “Chemical Genetics Uncovers Novel Inhibitors of Lignification” is summarized here by Peter Minorsky (ASPB and Mercy College) who writes the monthly On the Inside column for Plant Physiology.
Lignin is a phenolic polymer predominantly deposited in the plant’s secondary thickened cell walls together with cellulose and other matrix polysaccharides. By rendering the cell wall impermeable, lignin allows long distance transport of water and dissolved nutrients along the vascular system. A lignin-impregnated cell wall also constitutes a physical barrier against insects and pathogens. In addition, the mechanical strength it provides to the plant ensures stability, erect growth, and resistance to the negative pressure caused by transpiration. Thus, the emergence of lignin is considered a crucial evolutionary adaptation of plants for successful land colonization.
On the other hand, lignin is a major factor causing cell wall recalcitrance for industrial processing of lignocellulosic biomass. For example, the extraction of fermentable sugars for the production of biofuels and other bio-based chemicals is largely hampered by the presence of lignin. To improve downstream processing, the biomass needs to be pretreated using harsh physical and chemical procedures. These additional processing costs give bio-based products a commercial disadvantage compared with conventional fossil-based alternatives.
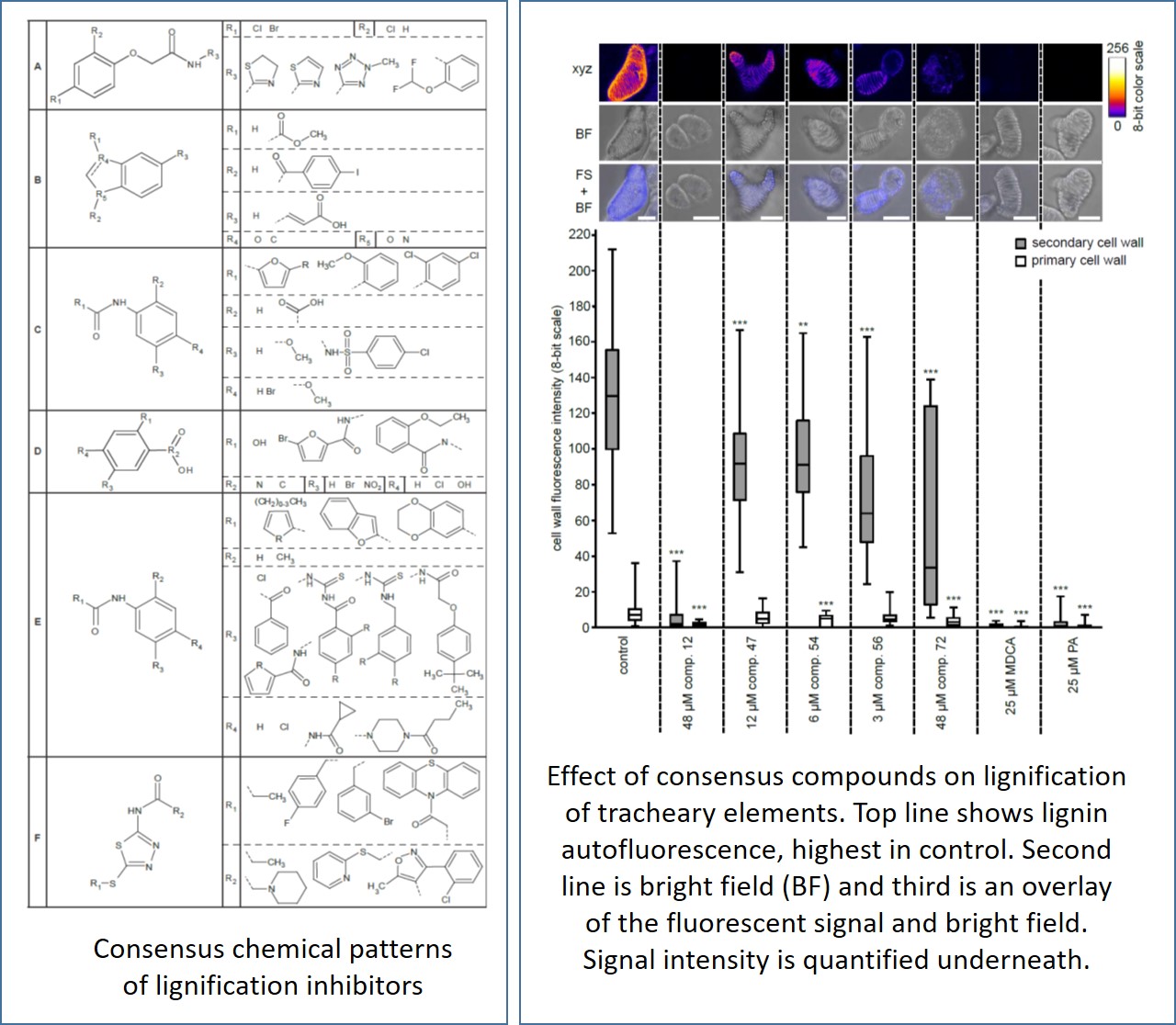
Van de Wouwer et al. performed a high-throughput screen of a diverse set of 10,000 synthetic molecules to identify inhibitors of the lignin biosynthetic pathway. Of the 73 positive hits, 39 putative inhibitors that caused major perturbations in the phenylpropanoid pathway were divided into five functional classes based on the phenolic profiles they provoked in seedlings. One representative compound of each functional class was then further subjected to a range of assays to quantify the extent of inhibition of lignin deposition and its effect on phenolic metabolism. This approach led to their discovery of p-iodobenzoic acid as a new inhibitor of cinnamate-4-hydroxylase, a key enzyme of the phenylpropanoid pathway which is responsible for synthesizing the building blocks of the lignin polymer.